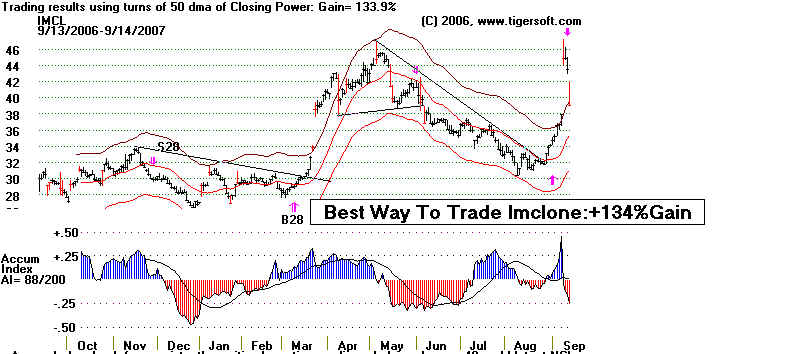
The Aftermath: IMCL Declines from 74 to 6 in Only 5
Months!
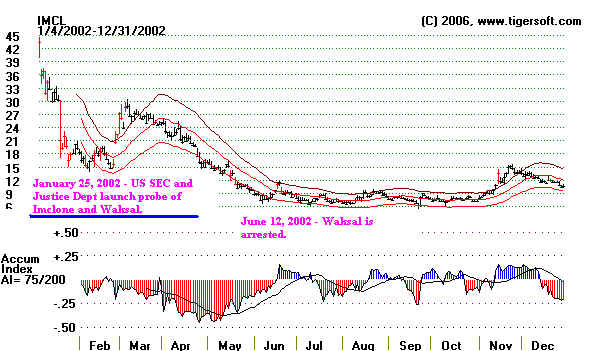
IMCLONE Time
Line: 2002-2003
Jan. 25, 2002 - U.S. Securities and Exchange Commission and the U.S. Justice
Department launch probe into ImClone and Waksal.
Feb. 14, 2002 - Waksal notifies the Securities and
Exchange Commission for the first time of 50 trades he made in ImClone stock going back as
far as 1992, transactions that should have been reported within months of their
execution.
May 22, 2002 - Samuel Waksal resigns as CEO of ImClone. Samuel's brother Harlan Waksal
takes over as CEO.
June 12, 2002 - Samuel Waksal is arrested on charges he illegally acted on inside information in attempting to sell ImClone shares, and
tipped family members to do the same.
Aug. 7, 2002 - Samuel Waksal, already charged with insider trading, is indicted for
obstruction of justice and bank fraud in a case that has rocked investors' confidence.
Waksal is accused of pledging ImClone securities he no longer owned to secure a $44
million loan and forging the signature of ImClone's general counsel to fool the bank into
believing he still owned the securities.
Aug 14, 2002 - ImClone sued Samuel Waksal, claiming Waksal ordered the destruction of
documents that may be important in a government investigation into the company.
Oct. 15, 2002 - Waksal entered a partial guilty plea to insider trading and obstruction
of justice charges.
March 3, 2003 - Already awaiting sentencing for insider trading, Waksal pleads guilty
to charges of evading tax on $15 million of art. He allegedly sent the art to an ImClone
manufacturing facility in New Jersey to avoid New York sales taxes and then had the
paintings re-directed to his Soho apartment.
March 11,2003 - Waksal agreed to partially settle a U.S. SEC suit by agreeing to be
barred from acting as an officer of any publicly traded company and paying back more than
$800,000 in insider trading profits.
March 31, 2003 - ImClone discloses that Waksal failed to pay income tax on the exercise
of certain stock options and that it will take a charge of $23.3 million to account for
the omission.
April 9, 2003 - The company says securities regulators are investigating ImClone's
failure to pay taxes on stock options exercised by Waksal and other officers and
directors.
June 4, 2003 - Martha Stewart charged with obstruction of justice in connection with an
investigation into the timing of her sale of ImClone stock.
June 10, 2003 - Samuel Waksal, the former head of Imclone Systems was sentenced to 87
months in prison and is ordered to pay $3 million in fines for tax evasion and insider
trading.
|